Unitary transformations are known to preserve observable properties such as electron density. The hybridization of atomic orbitals is a kind of mathematical operation known as a unitary transformation. What I found is if you consider electron densities, the pictures of electrons in the unhybridized atomic orbitals filled according to the aufbau principle, 2s^2 2p^2 for carbon for example, and in hybrid orbitals, one electron in each of the four sp^3 orbitals, are equivalent pictures. This is a question I have investigated in my research and published on. This is an excellent question and you have received some excellent replies already. The other rules apply also - Pauli exclusion (only 2 electrons per orbital) and Hund's rule (electrons fill degenerate orbitals one at a time first, then pair up).įor a good sophomore organic summary, see Jakob Fredlos' book.įor a higher level treatment, I recommend Anslyn and Dougherty. Once we have the molecular orbitals for a molecule, aufbau ideas generally DO apply: the low energy orbitals are filled and the higher energy orbitals are not. But the actual system doesn't need to see the mixing - it just does it. But even if we fully "believe" in hybridization, it only applies to atoms in molecules that are making bonds - individual isolated atoms won't hybridize! It is easier for us to "pre-mix" orbitals in the atom (hybridization) then to visualize how the native atomic orbitals would mix to make bonding orbitals. It is useful in this respect, but a lot of experimental data shows that hybridization is not a very good model. 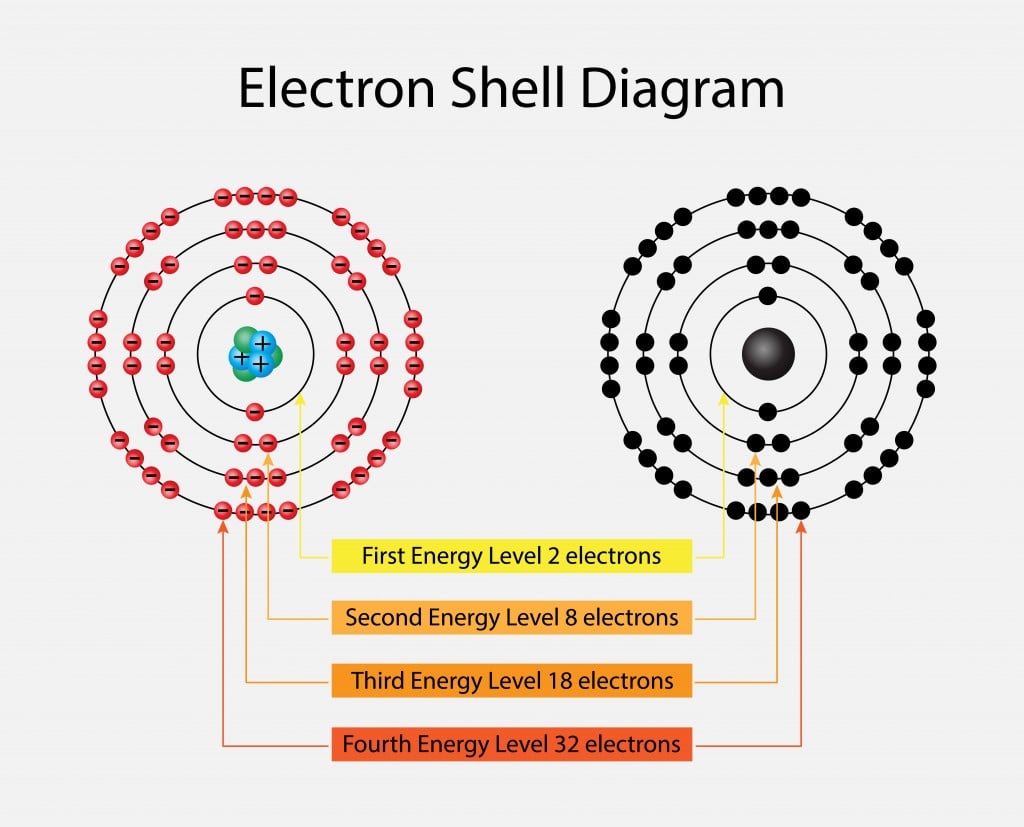
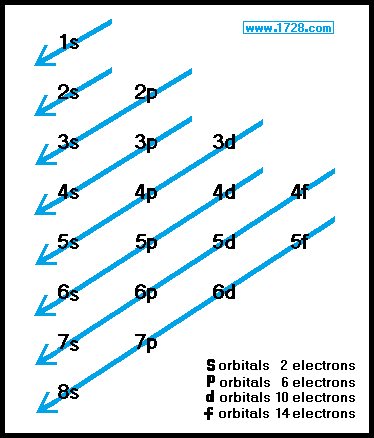
Hybridization is a model that allows us to better visualize the way bonds form. acdi-base theory) and develop new ones (molecular orbitals) as necessary to describe our observations more closely and to predict properties more accurately. We continue to observe, experiment and observe some more. However, it does not and was never intended to describe molecular orbitals or hybridization. Similarly, the Aufbau Principle serves as a good guideline for understanding the observations of elemental structure. Just as we know that the “Bohr Model” does not reflect the reality of atomic structure in many regards, it is still a useful starting point for atomic discusssions. The entire topic of hybridized orbitals is again an extension of previous theories into real observations. Related to your question, the Aufbau Principle applies only to ATOMS, not molecules. They are still subject to change, revision or disproving. At best, even “Laws” have simply been observed to make the best predictions for utility. First of all, always keep in mind that all scientifice theories are simply attempts to make some predictive sense from our observations. The application of the third principle can be seen in the electron configurations for the remaining second-row elements.Simply, no. When dealing with degenerate orbitals, such as p orbitals, one electron is placed in each degenerate orbital first, before electrons are paired up. The application of the first two principles can be seen in the electron configurations shown in (H, He, Li, and Be). In order for the orbital to accommodate two electrons, the electrons must have opposite spin states.ģ. For reasons that are beyond the scope of this course, electrons only have two possible spin states (designated by or ). To understand what “spin” means, we can imagine an electron spinning in space (although this is an oversimplified explanation of the term “spin”). Each orbital can accommodate a maximum of two electrons that have opposite spin. The lowest-energy orbital is filled first.Ģ. The order in which the orbitals are filled by electrons is determined by just three simple principles:ġ. Īs we move across the periodic table, starting with hydrogen, each element has one more electron than the element before it. Orbitals with the same energy level are called degenerate orbitals. After the 2s orbital, there are three 2p orbitals that are all equivalent in energy to one another. The 2s orbital has one node and is farther away form the nucleus it is therefore higher in energy than the 1s orbital. Electrons are lowest in energy when they occupy a 1s orbital, because the 1s orbital is closest to the nucleus and it has no nodes (the more nodes that an orbital has, the greater its energy). Our discussions will therefore focus primarily on these orbitals. These elements utilize the 1s orbital, the 2s orbital, and the three 2p orbitals. Most of the organic compounds that we will encounter will be composed of first- and second-row elements (H, C, N, and O). The energy of an electron depends on the type of orbital that it occupies.
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
